Sintering in the Powder Metallurgy Process
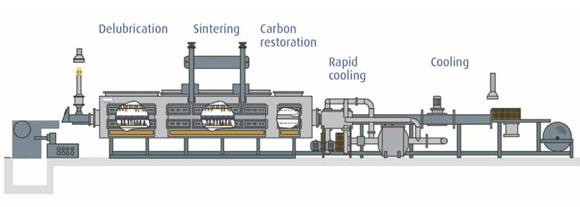
Sintering is a heat treatment applied to a powder compact in order to impart strength and integrity. The temperature used for sintering is below the melting point of the major constituent of the Powder Metallurgy material.
After compaction, neighbouring powder particles are held together by cold welds, which give the compact sufficient “green strength” to be handled. At sintering temperature, diffusion processes cause necks to form and grow at these contact points.

There are two necessary precursors before this “solid state sintering” mechanism can take place:-
- Removal of the pressing lubricant by evaporation and burning of the vapours
- Reduction of the surface oxides from the powder particles in the compact.
These steps and the sintering process itself are generally achieved in a single, continuous furnace by judicious choice and zoning of the furnace atmosphere and by using an appropriate temperature profile throughout the furnace.
Sinter hardening
Sintering furnaces are available that can apply accelerated cooling rates in the cooling zone and material grades have been developed that can transform to martensitic microstructures at these cooling rates. This process, together with a subsequent tempering treatment, is known as sintering hardening, a process that has emerged, in recent years, has a leading means of enhancing sintered strength.
Liquid Phase Sintering
Transient liquid phase sintering
In a compact that contains only iron powder particles, the solid state sintering process would generate some shrinkage of the compact as the sintering necks grow. However, a common practice with ferrous PM materials is to make an addition of fine copper powder to create a transient liquid phase during sintering.
At sintering temperature, the copper melts and then diffuses into the iron powder particles creating swelling. By careful selection of copper content, it is possible to balance this swelling against the natural shrinkage of the iron powder skeleton and provide a material that does not change in dimensions at all during sintering. The copper addition also provides a useful solid solution strengthening effect.
Permanent liquid phase sintering
For certain materials, such as cemented carbides or hardmetals, a sintering mechanism involving the generation of a permanent liquid phase is applied. This type of liquid phase sintering involves the use of an additive to the powder, which will melt before the matrix phase and which will often create a so-called binder phase. The process has three stages:
- Rearrangement
As the liquid melts, capillary action will pull the liquid into pores and also cause grains to rearrange into a more favourable packing arrangement - Solution-precipitation
In areas where capillary pressures are high, atoms will preferentially go into solution and then precipitate in areas of lower chemical potential where particles are not close or in contact. This is called contact flattening and densifies the system in a way similar to grain boundary diffusion in solid state sintering. Ostwald ripening will also occur where smaller particles will go into solution preferentially and precipitate on larger particles leading to densification. - Final densification
Densification of the solid skeletal network, liquid movement from efficiently packed regions into pores. For permanent liquid phase sintering to be practical, the major phase should be at least slightly soluble in the liquid phase and the “binder” additive should melt before any major sintering of the solid particulate network occurs, otherwise rearrangement of grains will not occur.
Next page: Properties of PM materials | Contents page
Download PM Review magazine











