2011 Hagen Symposium: Understanding the benefits of thermoanalysis for PM alloy development and the investigation of sintering processes
Dr. Christian Gierl of the Technical University Vienna, Austria, demonstrated in a presentation at the Hagen Symposium 2011, which took place in Hagen, Germany, November 24-25, the benefits of thermoanalytical methods for alloy development and for the investigation of sintering processes. Dr Georg Schlieper provides a summary of his presentation exclusively for ipmd.net.
The powder mix for compacting ferrous parts by coaxial pressing contains between 0.5 and 1.2% lubricating wax. This organic material is evaporated between 400 and 600°C. Continuous sintering furnaces are equipped with a separate lubricant burn-off zone where the atmosphere can be adapted to the requirements for a complete, residue-free removal of the lubricant. A slightly oxidising atmosphere supports the lubricant burn-off.
The next important reaction is degassing and oxide reduction. Chemical reactions are characterised by the free enthalpy of the compound. There is always an equilibrium concentration between the oxide and the reduced metal. The most widely used alloying elements in PM technology, Ni, Cu and Mo, are easier reduced than Fe. Therefore, for these alloys the reduction is dominated by the reactions of iron. Most common is the reduction of iron oxide with hydrogen to water vapour and with carbon to carbon monoxide. Gas reactions can best be studied with thermogravimetry (TG) and mass spectroscopy (MS). An example is given below.
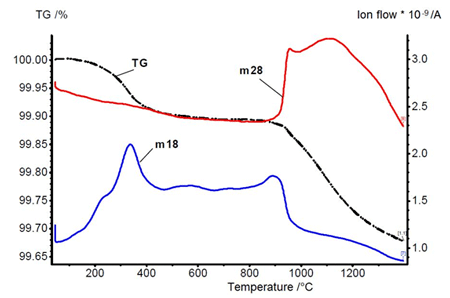
Thermogravimetry (TG) and mass spectroscopy (m18, m28) for Fe-4Ni-1.5Cu-0.5Mo-0.5C in hydrogen (courtesy C. Gierl)
The diagram shows the sample weight (TG), the formation of water vapour (m18) and carbon monoxide (m28) as a function of the temperature for a diffusion alloyed sintered steel. The test is able to distinguish between the reduction of oxides at the surface and in the interior of the powder particles. The first maximum for water vapour at 350°C is due to the reduction of iron oxide on the particle surfaces. Internal oxides need more time to reach the particle surface before they are reduced. The reduction temperature is above 900°C and the reaction product is always CO.
The thermodynamic properties of traditional PM alloys are the main reason for their commercial success. They can be sintered in practically all atmospheres that can be used for steel, either neutral or inert (nitrogen, vacuum) or reducing (nitrogen-hydrogen, endogas) of medium quality with respect to dew point and oxygen content.
The drawback of Ni, Cu, Mo is the high alloying cost. Particularly the price of molybdenum has increased in recent years. Fine nickel powder is frequently a concern because nickel is carcinogenic. The use of alloying elements for wrought steels such as Cr, Mn, V and Si has frequently been investigated in recent decades. Their oxides are much more stable than that of iron and therefore the reduction of surface oxides is no longer determined by the reduction of iron oxide. These elements also present higher requirements on the sintering atmospheres.
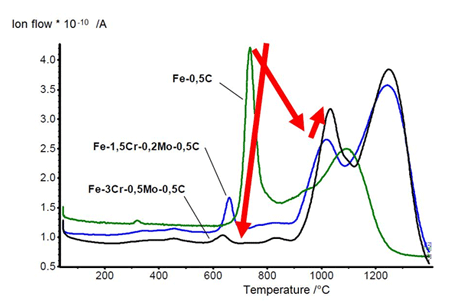
Chromium containing sintered steels exhibit significant changes in oxide reduction as shown in the mass spectroscopy for CO. The reduction of surface iron oxides, which is very strong for Fe-0.5C, is suppressed by chromium. Complex iron-chromium oxides are formed which are more stable than iron oxide and the reduction is shifted to temperatures above 1000°C.
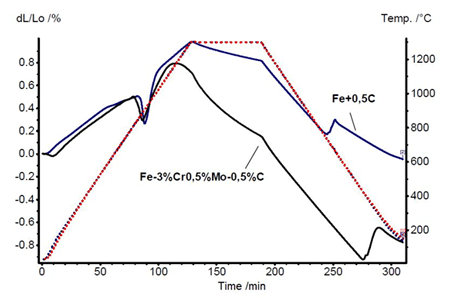
The formation of sinter necks, shrinkage during sintering and phase transformations can best be studied with dilatometer curves. Chromium alloyed steel has a much higher shrinkage than most standard alloys. In many PM steels, Cu is used to correct the shrinkage and thus achieve high dimensional precision. Parts producers are cautious as to whether PM steels containing Cr can meet tight dimensional tolerances to the same extent as the traditional alloys do.
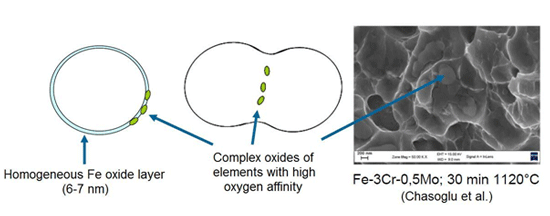
Powder metal alloys containing elements with a high oxygen affinity such as Cr, Mn and Si can be sintered thanks to an internal getter effect. The homogeneous iron oxide layer that covers the powder particles is approximately 6-7 nm thick. If the powder is prealloyed and the alloying elements are uniformly distributed in the material, the oxide layer protects the alloying elements from being oxidized, except for a small amount of complex oxides at the particle surface. Iron oxide is reduced in the early stage of sintering (~ 350°C). The complex oxides are integrated into the structure as sinter necks are formed. At high temperatures these oxides are reduced by carbon. This is associated with a slight loss of carbon.
Dr. Gierl concluded his presentation with a discussion of a complex Fe-Mn-Si master alloy for introducing Mn and Si as alloying elements in PM technology. The internal getter effect was confirmed, but it was also found that the formation of methane can lead to a substantial loss of carbon and oxidize the alloying elements.
News | Articles | Market reviews | Search directory | Subscribe to e-newsletter