2011 Hagen Symposium: The Powder Metallurgy of Refractory Metals
In his Skaupy speech Dr Gille gave a highly ambitious, scientifically in-depth, yet comprehensible lecture about two topics of refractory metal products – tantalum capacitors and WC-Co hardmetals.
Modern powder metallurgy started some 100 years ago with refractory metals, namely the production of tungsten filaments for light bulbs. Refractory metals such as tungsten, molybdenum, tantalum, niobium, rhenium could not be melted due to their high melting points and so sintering was the only way to transform them into technical products. Today modern melting technologies such as electron beam, plasma arc and scull melting are available, but powder metallurgy is still the primary processing technology for refractory metals.
Tantalum capacitors
By taking tantalum powders for capacitors as an example, Dr Gille demonstrated that sophisticated powder design can be the key to high performance properties of end products.
The rapid development of microelectronics since the 1970’s has not only led to miniature computer chips, but at the same time stimulated the improvement and miniaturisation of inactive circuit elements such as capacitors. Without these developments, the small size of today’s mobile phones, digital cameras and handheld computers would have been impossible.
Tantalum capacitors offer the highest performance of all known designs. The principle is based on tantalum and manganese oxide electrodes separated by a dielectric in the form of a tantalum oxide film. The capacitance of the system is determined by the area A and the thickness d of the dielectric.
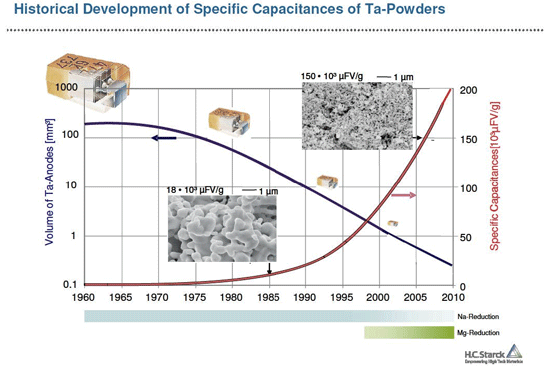
HC Starck developed a new patented process for making Ta powder with extremely high internal surfaces. The process is based on the reduction of Ta2O5 powder by magnesium vapour at 850 to 950°C. The resulting powder with an extremely fine spongy structure and internal surfaces of 2 to 5 m²/g is pressed and sintered to form the core of the capacitor. The internal surface of the sintered body is then oxidised in an electrochemical process and finally coated with the cathode material.
The powder production process can be conducted in a way as to control the pore size of the powder, which directly corresponds to the internal surface area.
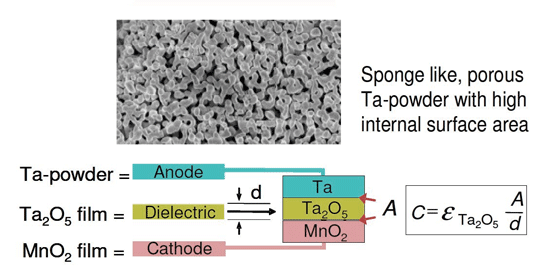
The function of Ta capacitors is based on Ta and MnO2 electrodes separated by a Ta2O5 film (courtesy HC Starck)
WC-Co Hardmetals
Hardmetals for high performance cutting applications are increasingly based on ultrafine and even nanoscale WC powders. With these powders it is possible to sinter microstructures of excellent homogeneity which combine extremely high hardness and strength characteristics.
The production of these ultrafine powders requires a thorough understanding of the chemical reactions for reducing tungsten containing compounds to pure tungsten powder. The degree of agglomeration must be low enough so that ultrafine WC particles are formed when the reduced W powder is carburized to WC powder of equivalent fineness.
Equally important as the quality of the powder is the sintering process. Dr Gille made some interesting statements about the sintering of hardmetals. He said that although hardmetal is the typical example for liquid phase sintering, most of the densification takes place in the solid state. For standard and coarse grained hardmetal the densification during solid state sintering is between 65 and 85% and for fine, ultrafine and nanoscale hardmetal it is 85 to 95%.
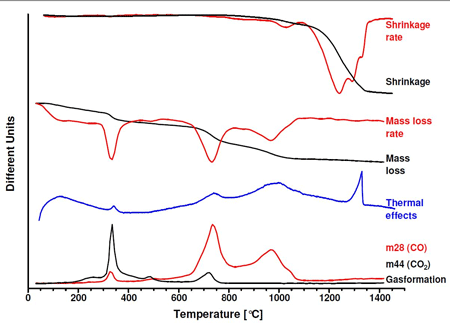
Besides the evaporation of wax, which plays an important role for processing granulated hardmetal powders, the reduction of surface oxides controls the densification processes. Thermoanalytical methods such as dilatometry, thermogravimetry, digital scanning calorimetry and mass spectroscopy can be used to determine the temperature ranges and quantify the effects. These experiments show that surface oxides of cobalt binder metal are typically reduced at 330°C, those of WC particles at approximately 750°C and dope carbides VC/Cr3C2 are reduced at 950°C.
Consideration must also be given to the amount of carbon which is lost in the reduction due to the Boudouard equilibrium between carbon, carbon monoxide and carbon dioxide. The carbon loss must be compensated by an appropriate addition of graphite to the powder mix. Gases formed in the reduction must be allowed to escape from the material before the porosity is closed due to shrinkage, otherwise there is a risk that trapped gases might form pores. This is particularly critical for ultrafine and nanoscale powders where shrinkage occurs at lower temperatures.
News | Articles | Market reviews | Search directory | Subscribe to e-newsletter